Abstract
Background: Early recurrence after first line therapy in patients with untreated follicular lymphoma (FL) has been shown to correlate with worse overall survival (Casulo et al., JCO 2015). Numerous clinical and molecular models have attempted to predict outcome and guide therapy. Lymphoma involvement of extra-nodal (EN) sites is better detected by fluorodeoxyglucose-positron emission tomography/computed tomography (FDG-PET/CT) than CT alone, but PET detected parameters are not part of the usual predictive indices. In a retrospective study of 613 cases of newly diagnosed FL grade 1-3a, we found that qualitative PET/CT abnormalities of spleen, skin/soft tissue, and bone, as well as involvement of ≥2 EN sites, predicted early clinical failure (St-Pierre et al., AJH 2019). We aimed to validate these findings with a separate cohort of patients with untreated high-tumor burden FL. Additionally, we aimed to evaluate the prognostic significance of a predictive index incorporating the PET factors that were validated in this study.
Methods: PET/CT images from 258 patients with newly diagnosed grade 1-3a FL included in the ECOG-ACRIN E2408 randomized phase II trial (Evens et al., Clin Cancer Res. 2020) were reviewed. Of these, 9 patients were excluded due to poor image quality. This study included untreated high-tumor burden FL patients who were treated with a bendamustine/rituximab backbone for induction followed by 2 years of rituximab-based maintenance. Location, pattern, and number of EN sites, as well as splenic involvement, were recorded. The primary endpoint was progression-free survival (PFS) and overall survival (OS). Time to event measures were estimated using the Kaplan and Meier method, where differences between groups were assessed using log-rank statistics. Multivariate analyses were performed using Cox-proportional hazards regression models. PET factors that had a statistically significant (p≤0.05) impact on PFS and/or OS were combined with statistically significant FLIPI and FLIPI-2 factors in this cohort to create a PET-based prognostic index for newly diagnosed FL.
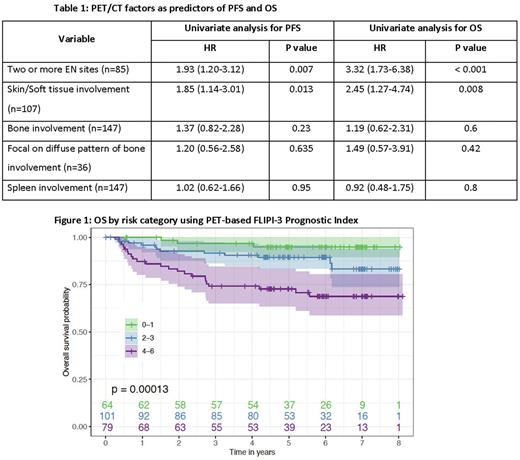
Results: Presence of ≥2 EN sites and skin/soft tissue involvement on PET/CT were validated as univariate predictors of PFS and OS. In a multivariate analysis with FLIPI-2 factors, presence of ≥2 EN sites on PET/CT was validated as an independent predictor of PFS (HR 1.69, p=0.045) and OS (HR 2.52, p=0.009). 2-year OS was 83% in the group with ≥2 EN sites compared with 94% in the group with 0-1 EN sites, and 84% in the group with skin/soft tissue involvement compared with 96% in the group with no skin/soft tissue involvement. In subset analyses, these PET factors were predictive of PFS and OS across all E2408 treatment arms. Bone and spleen involvement, contrary to the prior study, did not predict outcomes. Results are outlined in Table 1. Other factors that impacted PFS and/or OS in this cohort included ages > 60 years, longest diameter of any lymph node > 6 cm, hemoglobin < 12, and elevated β2-microglobulin (FLIPI-2). These factors were combined with the number of EN sites involved (0-1 vs 2+) and presence of skin/soft tissue involvement on PET/CT to form the PET-based FLIPI score. Each factor represents one point on a 6-point scale. A high-risk score of 4-6 using this index was associated with a 2-year PFS of 70% vs 87% in the intermediate-risk group (score 2-3), and 91% in the low-risk group (score 0-1) (p=0.00033). The 2-year OS was 82% in the high-risk group, compared with 93% in the intermediate-risk group, and 97% in the low-risk group (p=0.00013). These results are outlined in Figure 1. Using the same score without the two PET factors, the 2-year OS was 88% in the high-risk group (score 3-4), 90% in the intermediate-risk group (score 1-2), and 100% in the low-risk group (score 0) (p=0.0099).
Conclusions: In this validation study, we 1) confirm that involvement of ≥2 EN sites and skin/soft tissue involvement on qualitative PET/CT represent predictors of PFS and OS in newly diagnosed FL, while bone and spleen involvement were not validated. Furthermore, this was validated in a FL patient population treated with contemporary bendamustine-based induction therapy with rituximab maintenance. 2) The PET-FLIPI score combining these PET-derived factors with traditional FLIPI and FLIPI-2 provides a more robust prognostic index for PFS and OS in these patients. These results provide a readily available tool that may help guide future clinical trials.
Disclosures
Winter:Servier: Consultancy, Other: For Spouse; CVS/Caremark: Consultancy, Other: For Spouse; Astellas: Other: For Spouse, to University of Chicago, Research Funding; Rafael: Other: For Spouse, to University of Chicago, Research Funding; Forty Seven/Gilead: Other: For Spouse, to University of Chicago, Research Funding; Novartis: Consultancy, Other: for Spouse, to the University of Chicago, Research Funding; Daiichi Sankyo: Other: for Spouse, to the University of Chicago, Research Funding; Cellectis: Other: for Spouse, to the University of Chicago, Research Funding; Merck & Co., Inc.: Honoraria, Research Funding. Witzig:Karyopharm: Other: Clinical Trail Support; ADC Therapeutics: Membership on an entity's Board of Directors or advisory committees; Kura Oncology: Other: Clinical Trail Support; Curio Science: Honoraria. Kahl:AstraZeneca: Consultancy, Research Funding; ADT Therapeutics: Consultancy; Roche: Consultancy; Genentech: Consultancy, Research Funding; Abbvie: Consultancy, Research Funding; MEI: Consultancy; AcertaPharma: Consultancy; Pharmacyclics: Consultancy; Celgene/BMS: Consultancy, Research Funding; Beigene: Consultancy, Research Funding; Kite: Consultancy; Janssen: Consultancy; Incyte: Consultancy; Hutchmed: Consultancy, Research Funding; TG Therapeutics: Consultancy; Genmab: Consultancy; Seattle Genetics: Consultancy; Research To Practice: Speakers Bureau. Gordon:Zylem: Current equity holder in private company, Current equity holder in publicly-traded company, Patents & Royalties: Patent on nanoparticles for lymphoma therapy; Janssen: Other: DSMB; Ono Pharmaceuticals: Consultancy; BMS: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal